

|
| This project is collaborated between The Forsyth Institute (TFI) and The Institute for Genomic Research (TIGR), and is funded by National Institute of Dental and Craniofacial Research (NIDCR) |
|
|
|||||||||||||||||||||||||
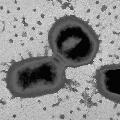
Integration of the information derived from the annotated sequence with the experimental data of the P. gingivalis research communityThe Forsyth investigators and consultants have particular interest in integrating the annotated sequence information with several areas of ongoing research. a) Integration of sequence information from previous studies and for other strains. As of September 25, 1996, there were 51 citations in GenBank for genes from Porphyromonas gingivalis. A web database will be constructed (see Aim 4) with links to those genes in GenBank and homologous genes in the TIGR P. gingivalis Database. Sequences from strain W83 will be checked for descrepancies with those previously published and an attempt will be made to resolve any sequence differences. Sequences for other strains will be aligned, and similarity values determined. b) Integration of sequence information for protease genes of P. gingivalis. A large number of protease genes have been found to date in P. gingivalis, additional homologs will be sought by homology searching, and the operon structure of the protease genes will be described. Consultant H.K. Kuramitsu has recently found that the major rgpA gene induces the expression of several other proteases. The genome organization of protease genes and their operon and promoter structures will be examined. c) Annotation of iron regulated genes. H. K Kuramitsu has recently identified a putative Fur-box in the promoter region of the rgpA protease gene. The Fur homolog in P. gingivalis will be identified as well as Fur-boxes in the promoter regions of other genes. d) Annotation of genes identified in ongoing IVET. In recent years studies of pathogen-host interactions have advanced significantly with the development of vectors allowing for the identification of virulence genes that are only expressed in animals during infection (Mahan et al.,1993; Camilli et al., 1994; Mahan et al., 1995 ). Consultants J.D. Hillman and A. Progulske-Fox are currently seeking to identify virulence genes using an IVET based strategy. A suicide vector has been constructed in which random P. gingivalis genomic fragments can be inserted upstream of a promoterless tetracycline resistance reporter gene. Transconjugants that contain, but do not express the reporter gene are identified by hybridization. Pools of such transconjugants are used to infect tetracycline-treated animals (mouse abscess model). Among strains that can establish an infection will be those with the tetracycline resistance reporter gene expressed from promoters which are induced in vivo but not in vitro. The unequivocal identification of these promoter sequences, and subsequently the genes they control, will be identified by searching the P. gingivalis genome sequence. Each gene found in this manner will be described in a page on the Forsyth P. gingivalis web site. e) Integration of genes sequences identified in B. thetaiotaomicron. A project to sequence genes from B. thetaiotaomicron is being carried out by John D’Elia, a graduate student in Consultant Abigail Salyer’s laboratory. To date more than thirty genes have been identified. A web database with a page for each B. thetaiotaomicron gene sequenced will be constructed with links to their GenBank entry and to homologous genes in the TIGR P. gingivalis Database. The results of the integration efforts in Aim 3 are made available on this web site. |
||||
|
This page is created and maintained by Drs. Margaret Duncan, Floyd Dewhirst, and Tsute Chen, Department of Molecular Genetics, The Forsyth Institute . Last modified on 02/20/2002 Copyright 2000, 2001, 2002 by The Forsyth Institute |